Module 5: The Synapse#
A synapse is a junction between two neurons (nerve cells) or between a neuron and another cell, such as muscle or a gland cell that allows them to communicate. It is the site where electrical or chemical signals are transmitted from one cell to another.
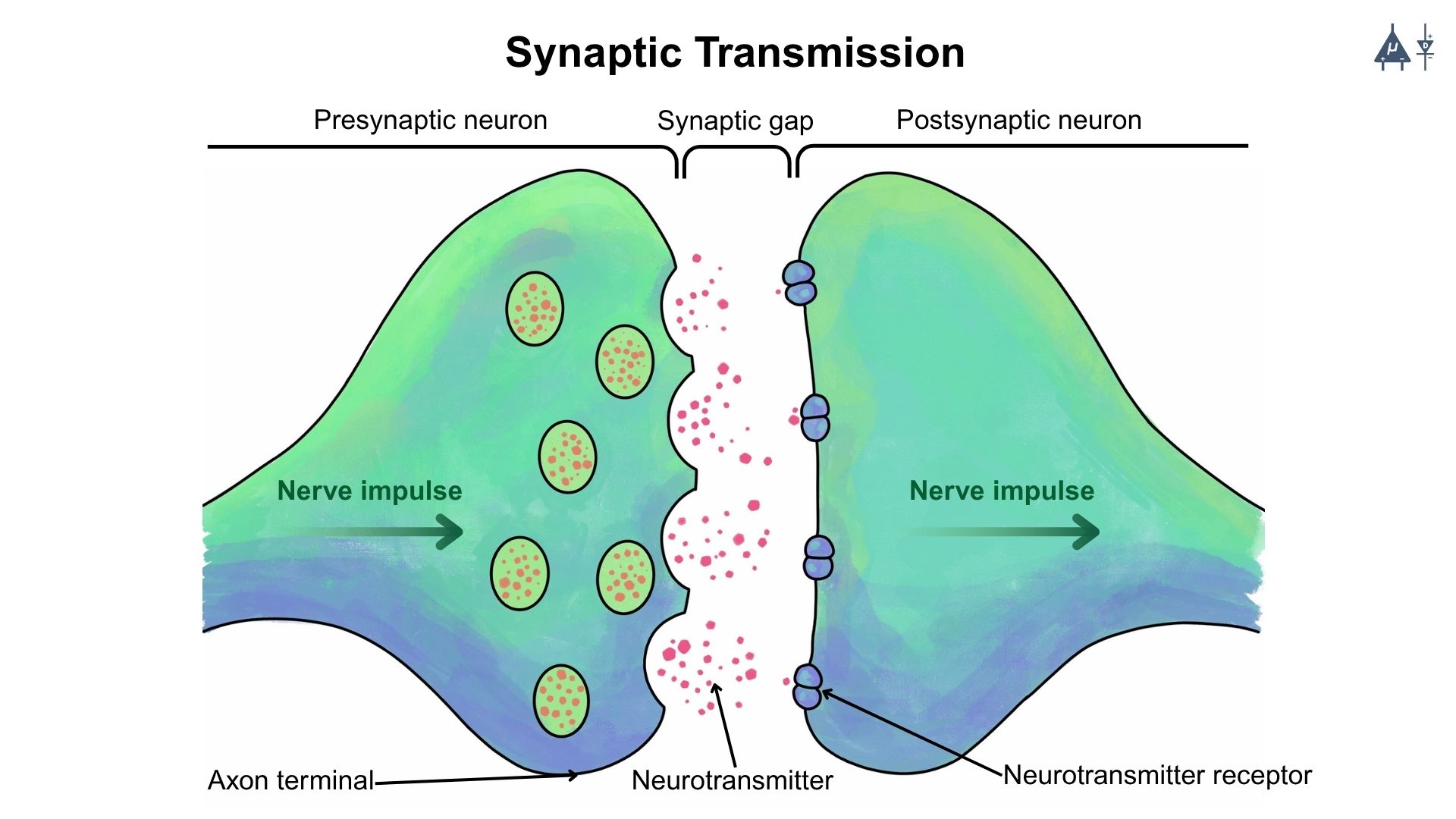
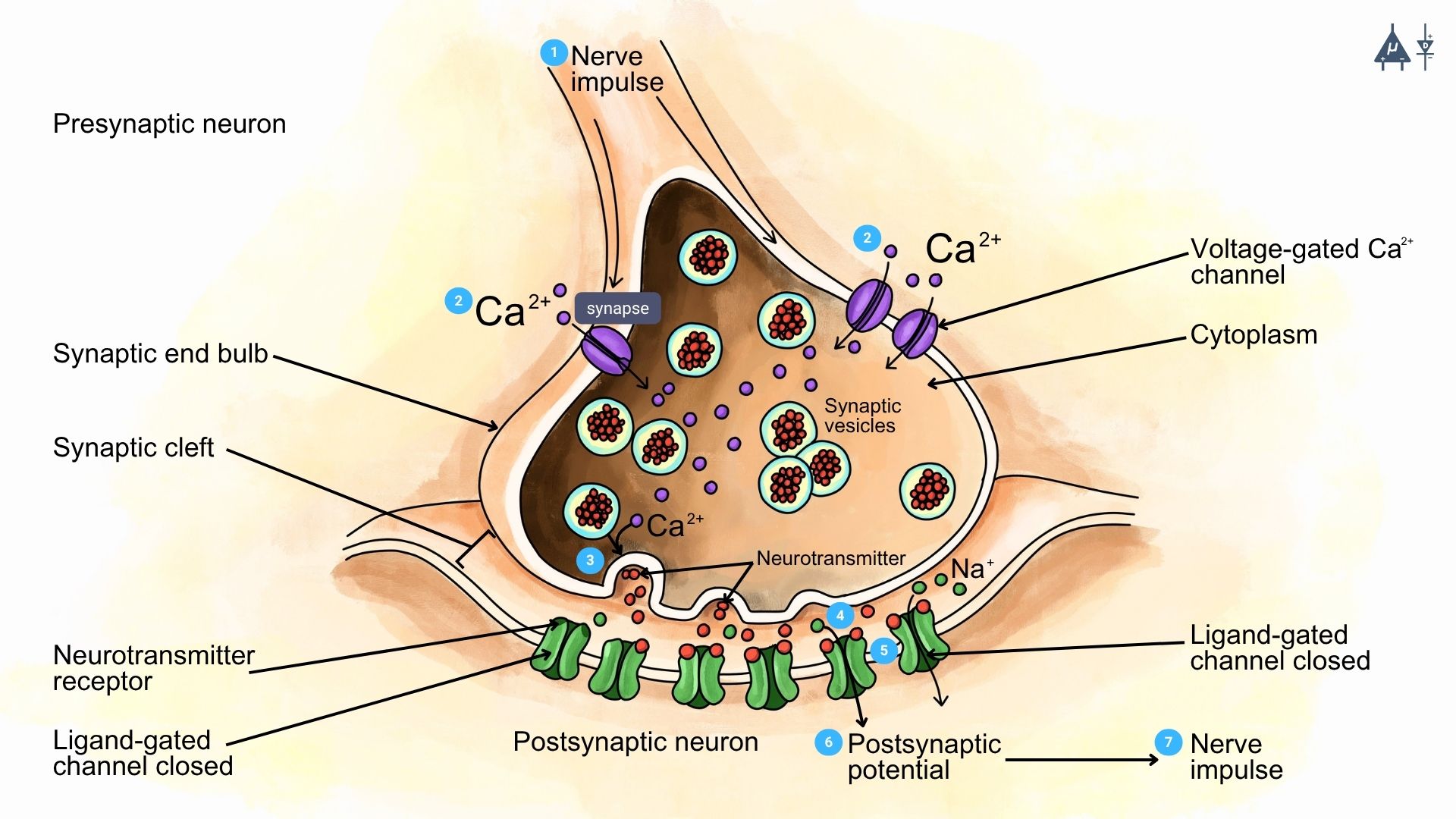
Schematic of synaptic transmission at a chemical synapse [9]#
5.1 Types of synapse#
There are generally 2 main types of synapse:
Electrical Synapse
Chemical Synapse
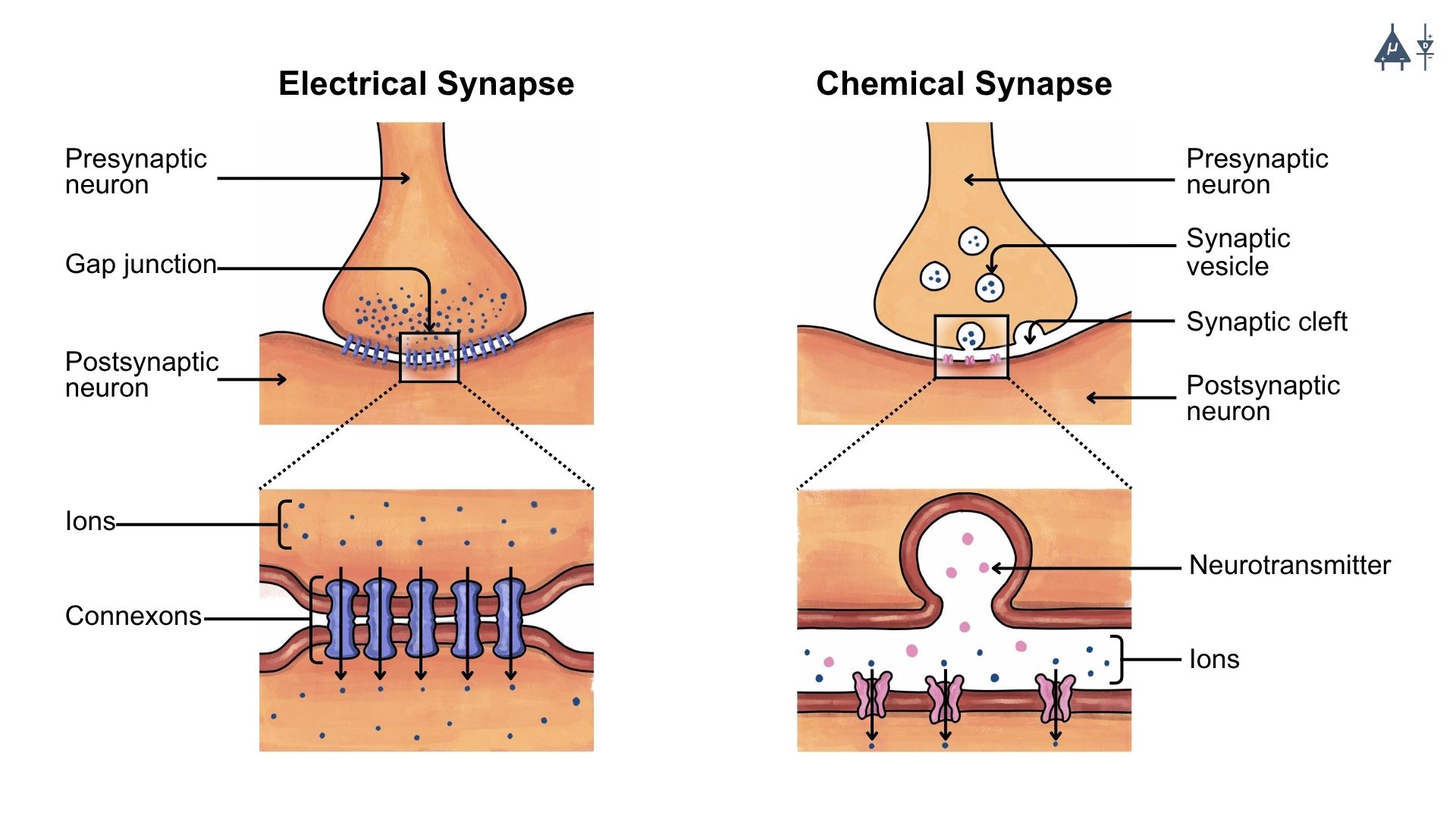
5.1.1 Electrical Synapse#
Electrical synapses allow direct communication between adjacent cells through specialized protein channels called gap junctions.
Structure of Electrical Synapse#
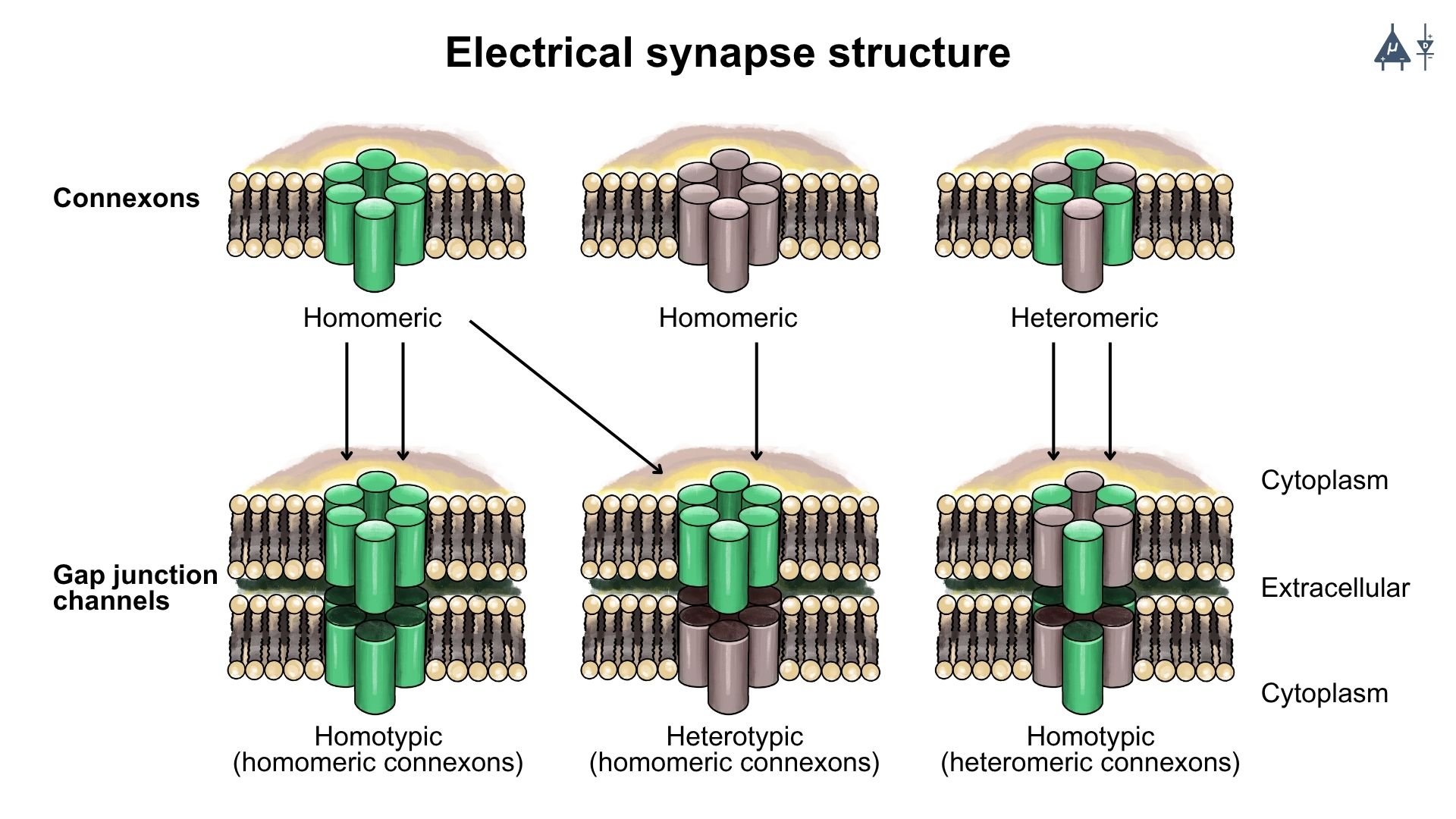
Electrical synapses are formed by gap junction channels that connect the cytoplasm of two neighboring cells.
Each gap junction channel is composed of two connexons, one from each cell.
Each connexon consists of six connexin proteins arranged in a circular pattern.
Mechanism of Electrical Synapse#
When an action potential (electrical signal) reaches the presynaptic terminal, it causes a rapid depolarization of the membrane.
This depolarization opens the gap junction channels, allowing ions to flow directly from the presynaptic cell to the postsynaptic cell.
This results in a nearly instantaneous transmission of the electrical signal, allowing for rapid communication between cells.
5.1.2 Chemical Synapse [6]#
Structure of Chemical Synapse#
Chemical synapse consists of a presynaptic neuron (which sends signals) and a postsynaptic neuron (which receives signal), separated by a synaptic cleft.
Mechanism of Chemical Synapse#
1. Arrival of Action Potential#
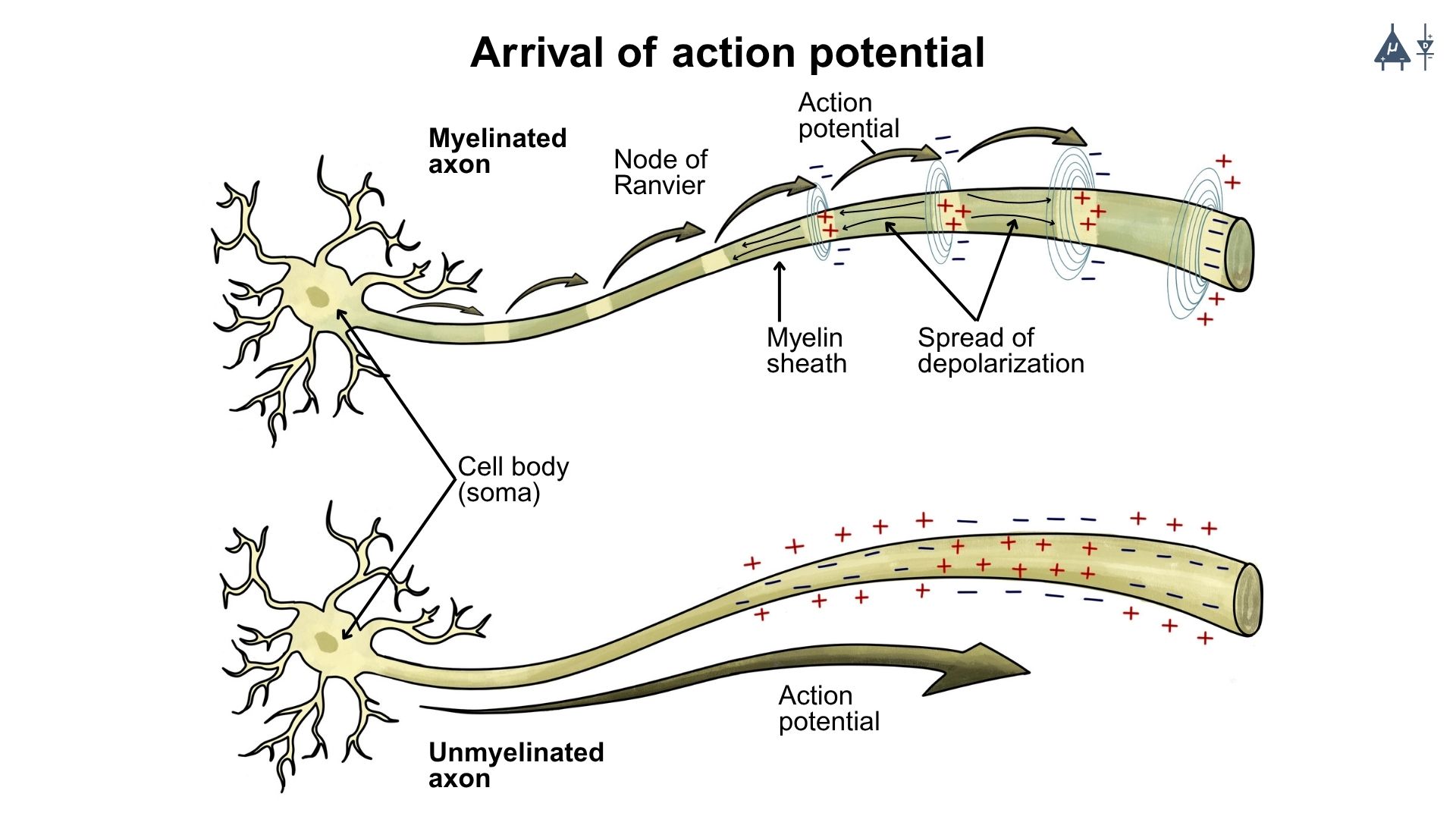
Arrival of action potential at the presynaptic terminal [11]#
When an action potential reaches the presynaptic terminal, it causes depolarization of the presynaptic membrane. This electrical signal initiates the movement of synaptic vesicles toward the presynaptic membrane near the synaptic cleft. These vesicles contain neurotransmitters, which function as chemical messenger molecules that transmit signals between neurons.
2. Opening of Voltage-Gated Calcium Channels#
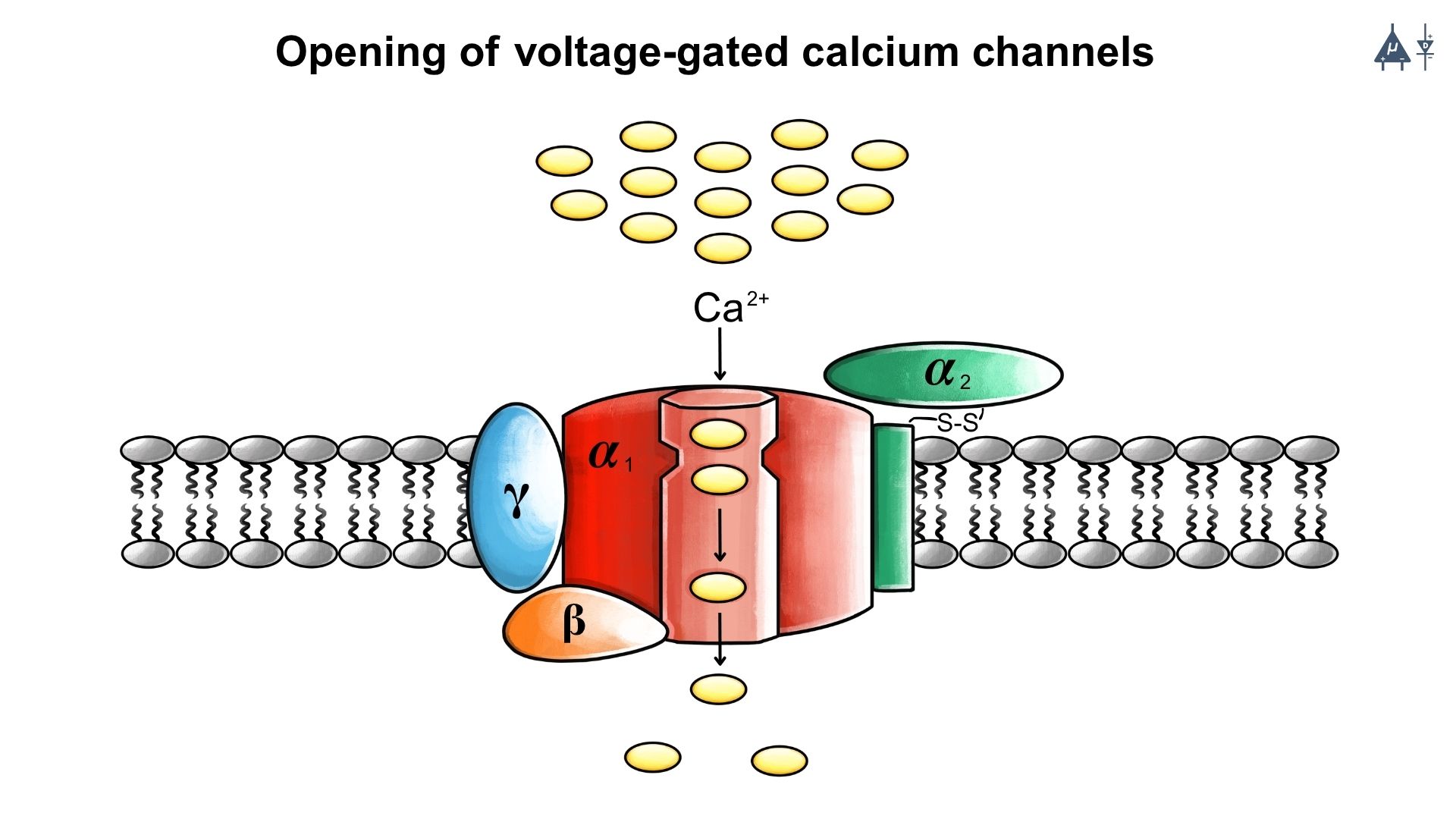
Opening of voltage-gated calcium channels and influx of calcium ions [12]#
The depolarization of the presynaptic membrane causes the opening of voltage-gated calcium (Ca²⁺) channels. As a result, calcium ions enter the presynaptic terminal from the extracellular fluid. The increase in intracellular calcium concentration triggers conformational changes in the proteins associated with synaptic vesicles. These changes prepare the vesicles for fusion with the presynaptic membrane.
3. Exocytosis of Neurotransmitters#
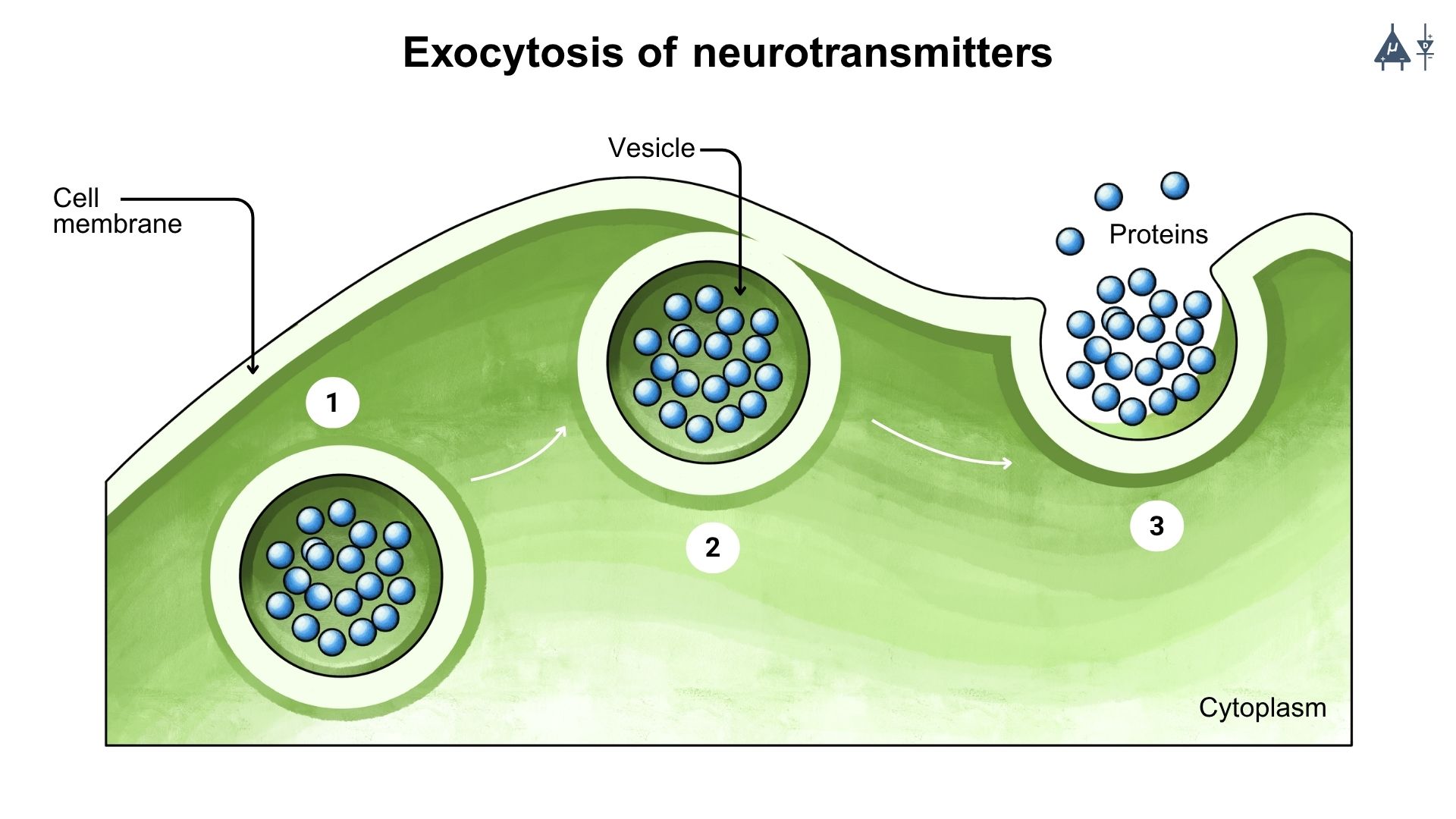
Exocytosis of neurotransmitters into the synaptic cleft [13]#
The influx of calcium ions causes the synaptic vesicles to fuse with the presynaptic membrane. This process is known as exocytosis. During exocytosis, the membrane of the vesicle merges with the plasma membrane and releases the stored neurotransmitters into the synaptic cleft.
4. Diffusion Across the Synaptic Cleft#
Once released, the neurotransmitters diffuse across the synaptic cleft and reach the postsynaptic membrane. The postsynaptic membrane contains specific receptors that recognize and bind to the neurotransmitters. These receptors are ligand-dependent, meaning they are activated when a neurotransmitter binds to them.
5.1.3 Types of Postsynaptic Receptors [7]#
Postsynaptic receptors can be broadly classified into ionotropic receptors and metabotropic receptors.
1. Ionotropic Receptors#
Ionotropic receptors are ligand-gated ion channels that open immediately when a neurotransmitter binds to them. The opening of these channels allows the movement of ions such as sodium, potassium, calcium, or chloride across the membrane, leading to rapid changes in the postsynaptic membrane potential. These responses are fast and short-lasting.
2. Metabotropic Receptors#
In contrast, metabotropic receptors are G-protein-coupled receptors (GPCRs). When a neurotransmitter binds to these receptors, it activates intracellular signaling pathways through secondary messengers. This process produces slower but more prolonged cellular responses compared to ionotropic receptors.
Cholinergic Receptors (Acetylcholine Mediated)
Cholinergic receptors are receptors that respond to the neurotransmitter acetylcholine. They are mainly classified into nicotinic and muscarinic receptors.
Nicotinic receptors are ligand gated ion channels. When acetylcholine binds to these receptors, conformational changes occur that open the ion channel and allow ions to enter the cell, producing a rapid excitatory response.
Muscarinic receptors, on the other hand, are G-protein coupled receptors. Binding of acetylcholine activates intracellular G-proteins that regulate different signaling pathways. The Gs protein stimulates adenylyl cyclase and increases cyclic AMP (cAMP) levels. The Gi protein inhibits adenylyl cyclase and decreases cAMP levels, while the Gq protein activates phospholipase C (PLC) leading to the production of IP₃ and DAG, which further mediate cellular responses.
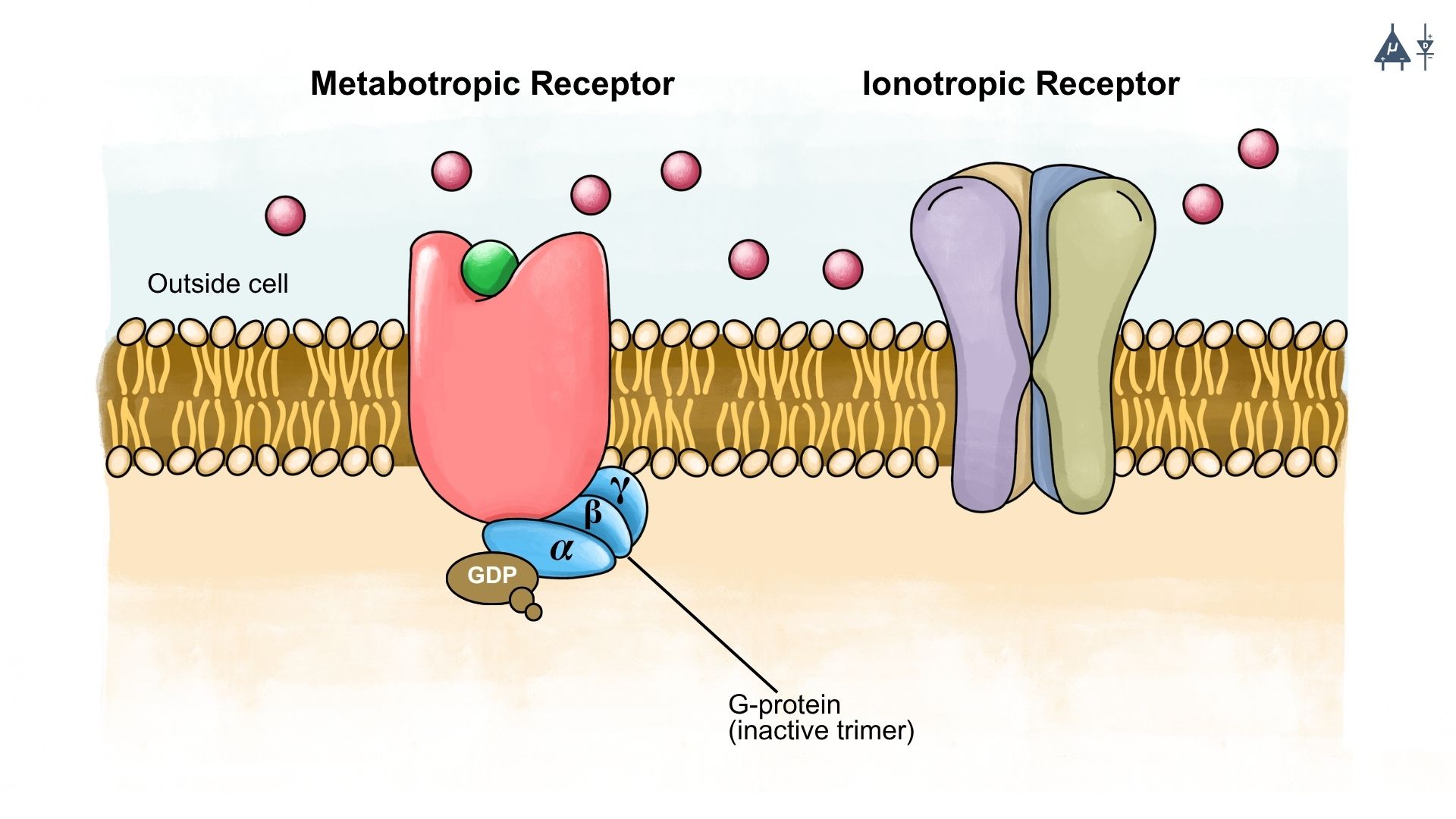
Types of postsynaptic receptors: Ionotropic (ligand-gated ion channels) and Metabotropic (G-protein-coupled receptors) [14]#
Note
For a very long time, scientists believed that electrical synapse is present in whole nervous system (DEVELOPED) but in reality it was chemical synapse which is more common.
As the axon of the presynaptic neuron gets closer to the synapse, it balloons out a little bit and this region is known as presynaptic terminal.
5.1.4 Key Differences#
Feature |
Electrical Synapse |
Chemical Synapse |
|---|---|---|
Mechanism |
Direct ion flow through gap junctions |
Release of neurotransmitters across a synaptic cleft |
Speed |
Fast (nearly instantaneous) |
Slower (due to chemical transmission) |
Directionality |
Bidirectional |
Unidirectional |
Signal Type |
Electrical signal |
Electrical + Chemical signal |
Examples |
Retina, heart, certain brain regions |
Most brain synapses, neuromuscular junctions |
Important Fact
In the developing nervous system, electrical synapse is more common.
In developed nervous system, chemical synapse is more common.
5.2 Fate of Neurotransmitters#
After neurotransmitters are released into the synaptic cleft, they do not remain there for long. Neurotransmitters present in the synaptic cleft generally follow three main pathways:
1. Reuptake#
Neurotransmitters are taken back into presynaptic neuron through specific transporter proteins present on the presynaptic membrane.
Once inside the neuron, some neurotransmitters are repackaged into synaptic vesicles for future release.
2. Enzymatic degradation#
Some neurotransmitters are broken down by specific enzymes present in the synaptic cleft.
This process helps terminate the action of neurotransmitters.
3. Diffusion#
Some neurotransmitters diffuse away from the synaptic cleft into the surrounding extracellular fluid.
This reduces their concentration at the synapse and helps stop synaptic transmission.
In addition, excess neurotransmitters may also be taken up by glial cells, which assist in clearing excess neurotransmitters from the synaptic space and maintaining synaptic homeostasis
5.3 EPSPs and IPSPs [2]#
When a chemical synapse releases neurotransmitters, they attach to receptors on the postsynaptic membrane. This changes the flow of ions into or out of the neuron, creating small electrical changes called postsynaptic potentials. These shifts slightly raise or lower the membrane potential from its usual resting level of about –70 mV. There are two main types: EPSPs and IPSPs.
Excitatory Postsynaptic Potentials (EPSPs)#
EPSPs occur when excitatory neurotransmitters-most commonly glutamate - open Na⁺ or Ca²⁺ channels.
Positive ions enter the neuron, causing depolarization (the membrane becomes less negative).
This brings the neuron closer to the threshold needed to fire an action potential.
The size of an EPSP depends on how much neurotransmitter is released, how many receptors are activated, how far the signal is from the axon hillock, and how much it decays along the membrane.
If enough EPSPs arrive together and push the membrane to about -55 mV at the axon hillock, an action potential is triggered.
Inhibitory Postsynaptic Potentials (IPSPs)#
IPSPs occur when inhibitory neurotransmitters-mainly GABA-open Cl⁻ channels (letting Cl⁻ in) or K⁺ channels (letting K⁺ out).
This adds more negative charge inside the neuron, causing hyperpolarization, which makes the neuron less likely to fire.
Like EPSPs, IPSPs are graded (Their size is not fixed - it depends on the strength of the input.) and weaken as they travel toward the axon hillock.
Important Points#
EPSPs and IPSPs are small changes from the –70 mV resting potential caused by shifts in ion permeability and basic electrical forces. (Module 2)
How far and how strongly they spread depends on membrane resistance (Rm), internal resistance (Ri), capacitance (Cm), the length constant (λ), and the time constant (τ). (Module 3)
When many EPSPs combine and reach threshold, the neuron fires an action potential. IPSPs work against this process, preventing the membrane from reaching threshold and reducing the chance of firing. (Module 4)
Synaptic transmission at the neuromuscular junction (NMJ) [16]#
5.4 Bernard Katz experiment [5]#
The Bernard Katz experiment, conducted in the mid-20th century by physiologist Bernard Katz and his colleagues, was pivotal in understanding synaptic transmission, particularly at the neuromuscular junction. Here’s a detailed overview of the experiment and its significance:
5.4.1 Overview of the Experiment#
1. Context#
Katz studied the neuromuscular junction in frogs, the place where motor neurons connect with muscle fibers. His goal was to figure out how a nerve signal triggers a muscle to contract.
2. Method#
Katz used electrophysiology to study how muscle cells respond electrically when a motor neuron activates them. He measured the postsynaptic potentials in the muscle fibers after stimulating the motor neuron and also observed how changing the calcium ion concentration affected these responses.
3. Findings#
Katz observed that when the presynaptic neuron releases neurotransmitters (mainly acetylcholine), the muscle fiber produces end-plate potentials (EPPs). End-plate potentials are the small electrical changes produced in muscle fiber when a motor neuron releases neurotransmitters at the neuromuscular junction.
He noticed that the strength of an EPP changes depending on how many neurotransmitter molecules are released.
When he lowered the calcium concentration in the surrounding solution, the EPPs became smaller, showing that calcium ions are essential for neurotransmitter release.
4. Quantal Release#
One of Katz’s major discoveries was the idea of quantal release, meaning neurotransmitters are released in small, fixed packets instead of continuously.
He confirmed this by studying miniature end-plate potentials (MEPPs), which occur even without any stimulation. Miniature end-plate potentials are tiny, spontaneous electrical signals seen in muscle fibers even when the motor neuron is not firing.
5.4.2 Significance:#
Bernard Katz studied the frog neuromuscular junction and discovered that neurotransmitters are released in discrete packets (quanta). Using electrophysiology, he showed that Calcium is essential for neurotransmitter release-lower calcium → smaller EPPs. His findings proved that synaptic transmission is quantized, not continuous, and earned him the Nobel Prize (1970).
5.5 SNARE Proteins [3]#
SNARE proteins are special proteins that help a neuron in releasing neurotransmitters. They are of 2 types:
v-SNAREs (vesicle SNAREs): These proteins are located on the synaptic vesicle membrane. The most common v-SNARE is synaptobrevin (also known as VAMP).
t-SNAREs (target SNAREs): These are found on the presynaptic membrane and interact with v-SNAREs to facilitate vesicle fusion. The main t-SNAREs include syntaxin and SNAP-25.
Mechanism of Action#
Vesicle Docking: v-SNAREs and t-SNAREs form a complex, bringing the vesicle close to the presynaptic membrane.
Fusion: Upon the arrival of an action potential, calcium ions enter the presynaptic terminal, triggering the conformational change in the SNARE complex that leads to the fusion of the vesicle with the membrane.
Neurotransmitter Release: Once the vesicle fuses with the membrane, neurotransmitters are released into the synaptic cleft.
5.6 Synaptic summation [4]#
Synaptic summation is the way a neuron adds up all the EPSPs and IPSPs it receives from thousands of synapses to decide whether it should fire an action potential. Since each EPSP or IPSP is very small (around 0.2–1 mV), a single synapse is never enough to make the neuron fire. Instead, the neuron relies on the combined effect of many inputs.
5.6.1 Types of synaptic summation#
There are two main types of summation:
1. Temporal Summation#
Temporal summation happens when several EPSPs or IPSPs arrive one after another in quick succession at the same synapse.
If they occur close together in time, they add up because the membrane hasn’t returned fully to its resting level.
This process depends heavily on the time constant (τ):
a. A longer τ means EPSPs last longer → easier to add up
b. A shorter τ means EPSPs fade quickly → harder to summate
2. Spatial Summation#
Spatial summation occurs when EPSPs or IPSPs arrive at the same time from different synapses located on different parts of the dendrites.
The inputs from many presynaptic neurons meet at the axon hillock and combine there.
It depends on the length constant (λ):
a. A larger λ allows EPSPs to travel farther with less decay
b. A smaller λ causes EPSPs to die out before reaching the hillock
5.6.2 Interaction of EPSPs and IPSPs#
Neurons constantly receive both excitatory and inhibitory signals.
EPSPs make the cell more positive → increase chance of firing
IPSPs make the cell more negative → reduce chance of firing
At the axon hillock, the neuron effectively performs a calculation:
Net input = total EPSPs - total IPSPs
If this net depolarization reaches about -55 mV → the neuron fires an action potential.
If inhibition outweighs excitation → no action potential occurs. This is the basic way neurons compute information.
5.6.3 How Synaptic Summation Generates Biopotential Signals (EEG, ECG, EMG, and EOG)#
Biopotential signals measured from the body like EEG, ECG, EMG, and EOG are not recordings of single neurons or single action potentials. Instead, these signals come from the summed postsynaptic potentials (EPSPs + IPSPs) of thousands to millions of cells firing together. This combined electrical activity creates voltage changes large enough to be detected by electrodes on the skin or scalp.
5.7 Additional info (Toxins and Their Effects)#
5.7.1 Botulinum Toxin (Botox)#
Mechanism: Botox is produced by the bacterium Clostridium botulinum and works by cleaving specific SNARE proteins (primarily SNAP-25).
Effects: This prevents vesicle fusion and neurotransmitter release, leading to muscle paralysis. Botox is used therapeutically for conditions like muscle spasms and cosmetics procedures by temporarily paralyzing muscles and by reducing the wrinkles and fine lines on skin.
5.7.2 Tetanus Toxin#
Mechanism: Produced by Clostridium tetani, tetanus toxin also targets SNARE proteins, specifically synaptobrevin.
Effects: This toxin inhibits the release of neurotransmitters that mediate inhibitory signals in the nervous system, leading to excessive muscle contraction and spasms (tetanus). It causes a severe and potentially life-threatening condition.
5.7.3 Alpha-Bungarotoxin#
Mechanism: Found in the venom of the banded krait snake, alpha-bungarotoxin binds irreversibly to nicotinic acetylcholine receptors at the neuromuscular junction.
Effects: This blocks the binding of acetylcholine, preventing muscle contraction and leading to paralysis.
5.7.4 Curare#
Mechanism: Curare is a plant-derived toxin that also binds to nicotinic acetylcholine receptors, but it does so reversibly.
Effects: Like alpha-bungarotoxin, curare prevents acetylcholine from activating muscle contraction, resulting in muscle paralysis. It has been used historically as a poison on arrows and darts.
5.7.5 Tetrodotoxin (TTX)#
Mechanism: TTX is found in pufferfish and works by blocking voltage-gated sodium channels on neurons.
Effects: By preventing sodium influx, TTX inhibits action potential generation and propagation, leading to paralysis and potentially death if ingested in sufficient quantities.
5.7.6 Saxitoxin#
Mechanism: Saxitoxin is produced by certain marine dinoflagellates and also blocks voltage-gated sodium channels.
Effects: Similar to TTX, saxitoxin causes paralysis by preventing action potentials.
5.7.7 Gabapentin and Pregabalin#
Mechanism: Gabapentin and pregabalin inhibit voltage-gated calcium (Ca²⁺) channels in presynaptic neurons, reducing calcium influx.
Effects: Reduced calcium entry leads to decreased neurotransmitter release from the presynaptic terminal.
Clinical Use: These drugs are commonly used as antiepileptic medications and are also used in the treatment of neuropathic pain.
Bonus Info
Do rusty nails actually cause tetanus?
Rusty nails themselves do not directly cause tetanus. The bacteria that lead to tetanus, Clostridium tetani, are commonly found in soil, animal feces, and dust. While rusty nails can create deep puncture wounds that allow these bacteria to enter the body, it’s the bacteria, not the rust, that poses the risk. If a wound becomes contaminated with C. tetani, tetanus can develop, which is why it’s important to keep wounds clean and up to date on vaccinations.
5.8 Summary#
In module 5, we explored how synapses enable communication between neurons, the difference between electrical and chemical synapses, how neurotransmitters create EPSPs and IPSPs, and how neurons add these signals through synaptic summation. We also learned about quantal release, the role of SNARE proteins, and how toxins affect synaptic function. All of this gives us the foundation to understand how large groups of neurons generate measurable electrical activity. In the next module, we will build on this knowledge and study biopotential signals such as EEG, EMG, ECG, and EOG how they are produced, how we record them, and what they tell us about the body.
5.9 References#
Pal, G. (2019). Comprehensive Textbook of Medical Physiology: Two Volume Set (2nd ed.). JP Medical.
https://www.simplypsychology.org/synaptic-transmission-a-level-psychology.html
https://colorado.pressbooks.pub/neuroscience/chapter/electrical-synapses/
https://antranik.org/synaptic-transmission-by-somatic-motorneurons/